QS-21 GH is a saponin-based adjuvant designed to support the development of vaccines for global health initiatives. Developed through our proprietary Q-SAP® technology platform, it offers equal biological activity than QS-21 INFINITY and scalable supply, ensuring confidence from preclinical studies to large-scale production.
Key Characteristics
- High Consistency: highly purified fraction of immunostimulatory saponins, consisting primarily of the QS-21 family.
- Form: Delivered as a lyophilized powder for ease of handling and storage.
- Validated Profile: Characterized by HPLC and LC-MS. Biological activity equals the one from legacy QS-21
- Available for Research: Samples are readily available for evaluation.
Specific antibody IgG2 titer induced by immunization with H1N1 HA, QS-21 GH + BECC470.
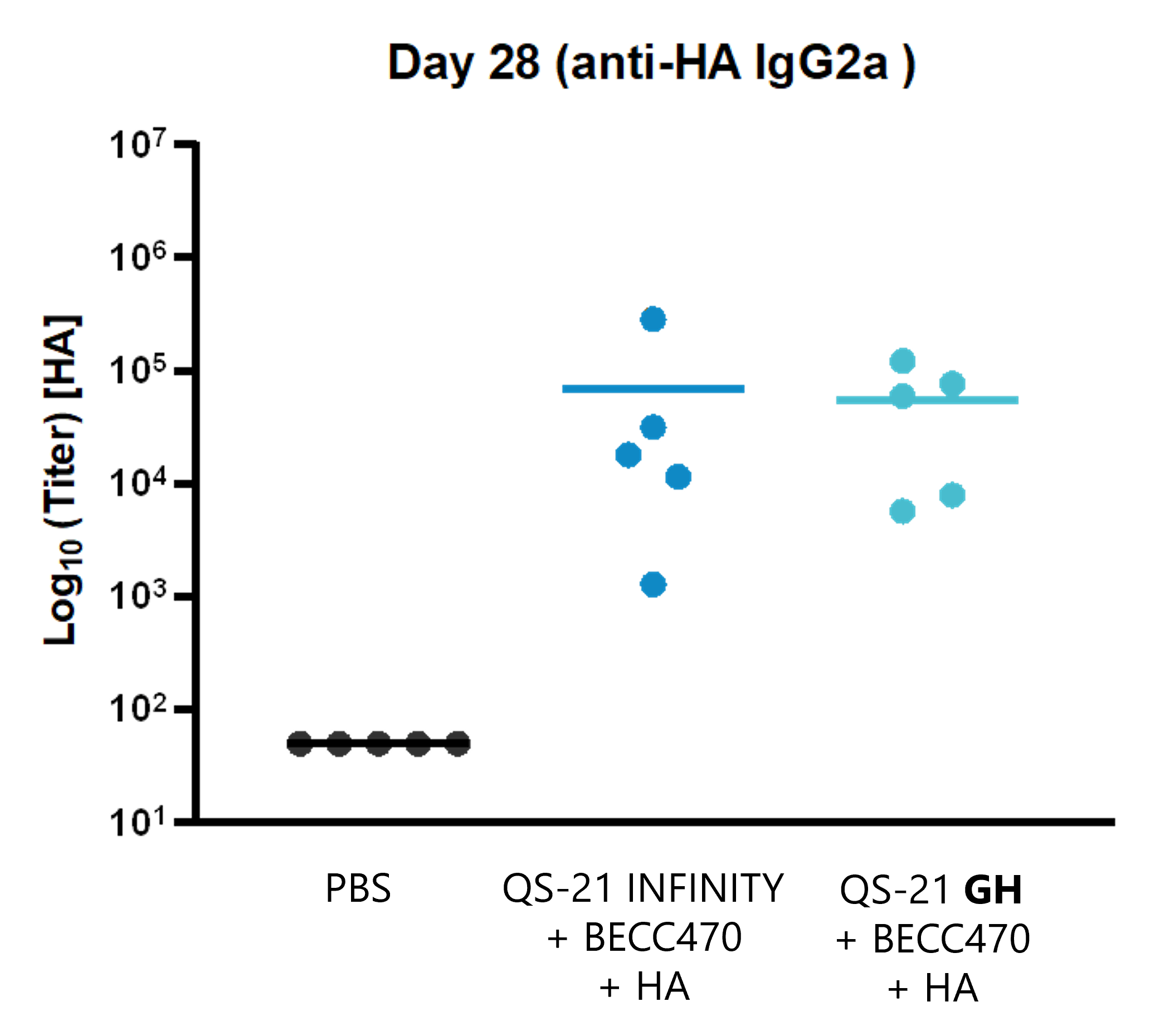
- QS-21 GH combined with a TLR4 agonist in a Liposome induces higher IgG2 antibody titer than the antigen alone.
- QS-21 GH combined with a TLR4 agonist in a Liposome induced equal IgG2 antibodies titer than QS-21 INFINITY.
Survival rate in a vaccine against Chagas Disease in mice
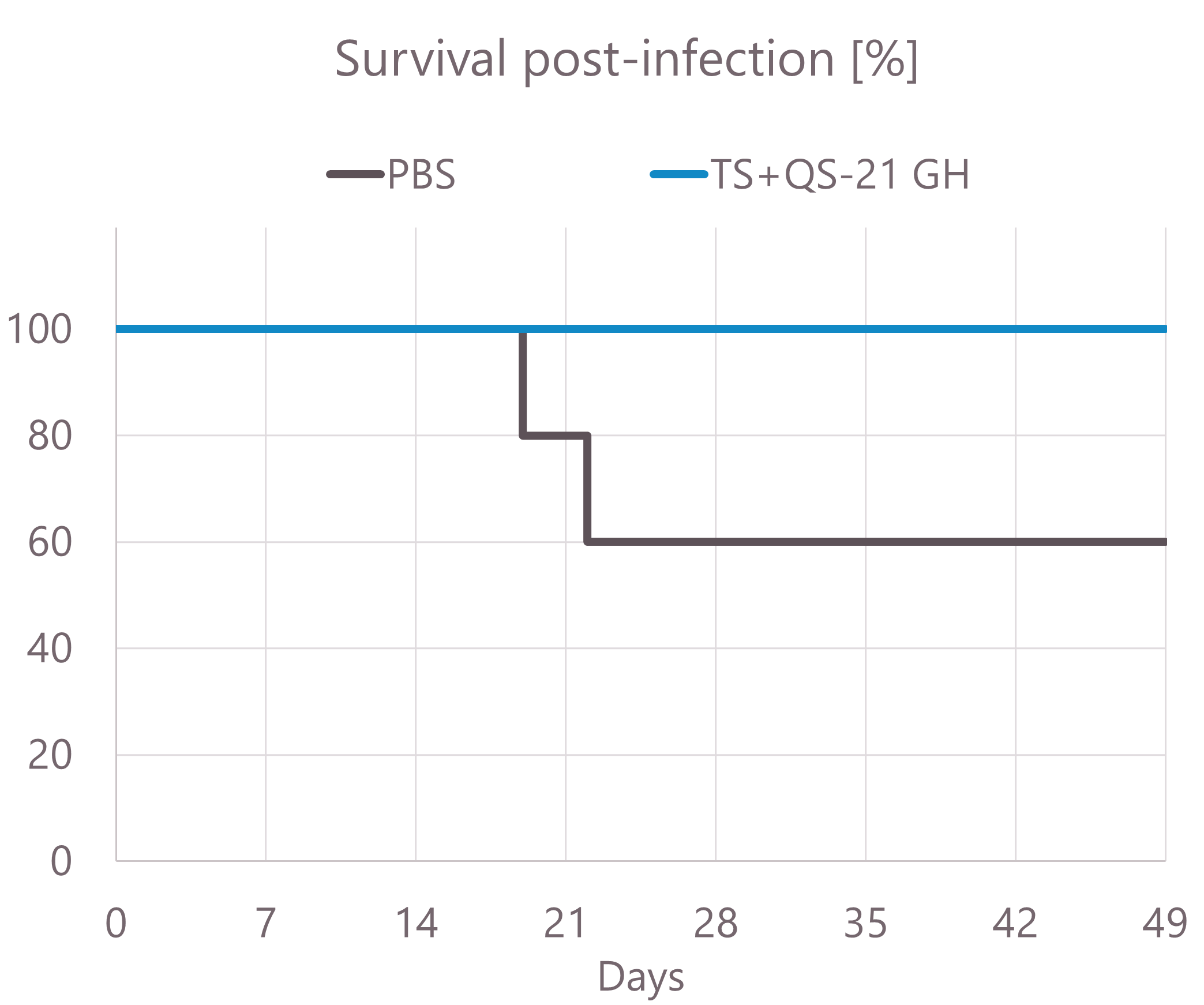
- The survival of the TS-QS-21 GH vaccinated group was 100% unlike 60% for the non-vaccinated group at day 22.

Partnership with SPI Pharma
Our strategic partner, SPI Pharma, facilitates access to samples for preclinical studies. With a strong focus on pharmaceutical innovation, SPI Pharma supports your R&D needs with fast and reliable sample delivery.

Formulation Support
The Q-VANT Biosciences team brings extensive expertise in vaccine formulation using saponin-based adjuvants. We offer guidance and technical insight to help you unlock the full potential of your research and development programs.

Why QS-21 Global Health?
- Secure Supply Chain: A reliable, scalable, and sustainable source of saponin-based adjuvant.
- Affordability: the cost of QS-21 GH enables vaccines against Malaria, Tuberculosis, Polio,…
- Open Access: QS-21 GH is IP free and openly accessible to all vaccine developers and manufacturers.
- Formulation-Ready: Lyophilized and quality-assured—ready to integrate into your vaccine development pipeline.
Request a Sample
Explore the future of vaccine adjuvants. Contact us to request your sample and connect with our scientific team.