Lyme disease is a tick-borne infection caused by Borrelia burgdorferi that leads to multisystem illness. Vaccine strategies use recombinant proteins with adjuvants to enhance immunity and broaden protection against diverse strains.
Lyme disease, caused by Borrelia burgdorferi, intracellular facultative bacteria and transmitted via Ixodes ticks. The development of an effective vaccine faces several challenges, including the induction of protective immunity, long-lasting immune memory and coverage against diverse Borrelia strains. Saponin-based adjuvants—particularly QS-21 can enhance immunogenicity and potentially broaden protective coverage.
Ma et al. (1994) evaluated the impact of adjuvants on immunogenicity in mice. They reported that QS-21-adjuvanted vaccines elicited higher antibody titers and more durable responses compared to alum.
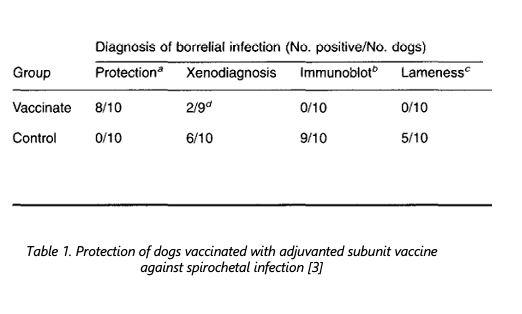
Saponin adjuvants were evaluated in the 1990s in the development of canine Lyme disease vaccines [1, 2] demonstrated that IgG1 and IgG2 antibody responses were four and eight times higher, respectively, in vaccines that contained QS21 and the recombinant Borrelia burgdorferi outer surface proteins OspA and OspB than in those that did not [3]. Dogs immunized were protected from Lyme disease upon challenge. Follow-up studies [4] confirmed the safety, efficacy, and immunogenicity of recombinant Osp A and B vaccines in dogs, where saponin-based adjuvants helped produce robust protection without unacceptable side effects.
References
1.Ma, J. et al., 1994. Impact of the saponin adjuvant QS-21 and aluminium hydroxide on the immunogenicity of recombinant OspA and OspB of Borrelia burgdorferi. Vaccine, 12(10), pp. 925-932.
2.Coughlin, R. T. et al., 1995. Protection of Dogs from Lyme Disease with a Vaccine Containing Outer Surface Protein (Osp) A, OspB, and the Saponin Adjuvant QS21. The Journal of Infectious diseases, 171(4), pp. 1049-1052.
3.Ma, J. et al., 1995. Characterization of Canine Humoral Immune Responses to Outer Surface Protein Subunit Vaccines and to Natural Infection by Lyme Disease Spirochetes. The Journal of Infectious Diseases, 171(4), pp. 909-915.
4.Ma, J. et al., 1996. Safety, efficacy, and immunogenicity of a recombinant Osp subunit canine Lyme disease vaccine. Vaccine, 14(14), pp. 1366-1374.