Equine vaccines often include saponin adjuvants to enhance both antibody and cell-mediated responses, offering durable protection against diseases such as anthrax, influenza, and strangles.
The adjuvant effect of saponin was first observed in 1925 by Gaston Ramon during diphtheria immunization studies in horses. In equine vaccines, saponin adjuvants have been utilized to enhance immunity against various diseases because it offers several advantages, including the induction of long-lasting immunity and the ability to stimulate both antibody and cell-mediated responses.
The use of saponins as adjuvant in spore vaccines also known as “Sterne Vaccine” presents a long history of effectiveness against anthrax [1].
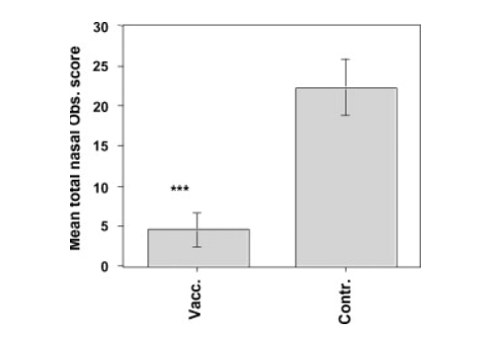
Ponies immunized with a vaccine adjuvanted against Equine Influenza showed higher levels of particular antibodies [2].
Adjuvanted vaccine against Streptococcus equi offered some degree of protection against strangles in ponies [3].
Soluble rVP2 vaccine adjuvanted with saponin against African Horse Sickness induced a complete protection [4].
Two injections produced total protection against the disease complex (EHV-2+ Rhodococcus equi) in most of the treated animals [5].
Horses who receive three doses of the basic vaccine are expected to have serum antibody titers that indicate they will be protected against tetanus for more than three years [6]
References
1.Sterne, M. & Robinson, E. M., 1939. The use of Saponin SPore vaccine for Inoculation against Anthrax in South Africa. Journal of Veterinary Science and Animal Industry, 12(2).
2.Paillot, R., Grimmett, H., Elton, D. & Daly, J. M., 2008. Protection, systemic IFN$\gamma$, and antibody responses induced by an ISCOM-based vaccine against a recent equine influenza virus in its natural host. Veterinary Research, 39(3).
3.Waller, A. et al., 2007. Vaccination of horses against strangles using recombinant antigens from Streptococcus equi. Vaccine, 25(18), pp. 3629-3635.
4.Scanlen, M., Paweska, J., Verschoor, J. & van Dijk, A., 2002. The protective efficacy of a recombinant VP2-based African horsesickness subunit vaccine candidate is determined by adjuvant. Vaccine, 20(7-8), pp. 1079-1088.
5.Nordengrahn, A. et al., 1996. Equine herpesvirus type 2 (EHV-2) as a predisposing factor for Rhodococcus equi pneumonia in foals: prevention of the bifactorial disease with EHV-2 immunostimulating complexes. Veterinary Microbiology, 51(1-2), pp. 55-68.
6.Kendall, A. et al., 2015. Duration of tetanus immunoglobulin G titres following basic immunisation of horses. Equine Veterinary Journal, 48(6), pp. 710-713.